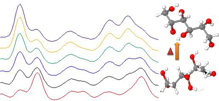
Raman spectroscopy is becoming the state-of-the-art in a variety of pharmaceutical applications, from high-throughput screening in drug discovery to manufacturing and quality control. Because of the technique’s inherent non-contact manner, Raman measurements can be obtained in a variety of environments and application areas while maintaining sterility and eliminating contamination.